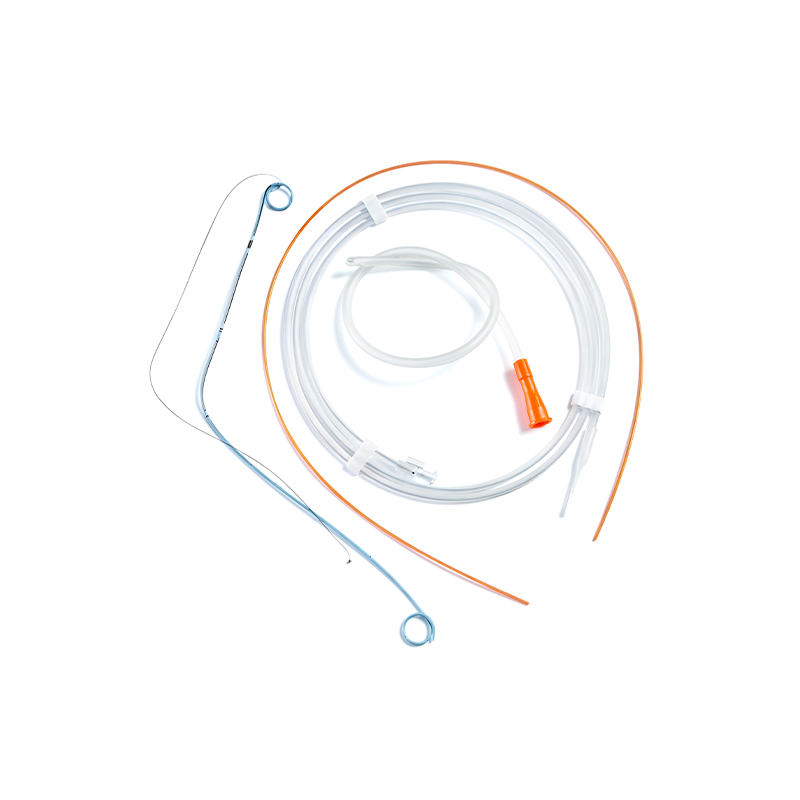
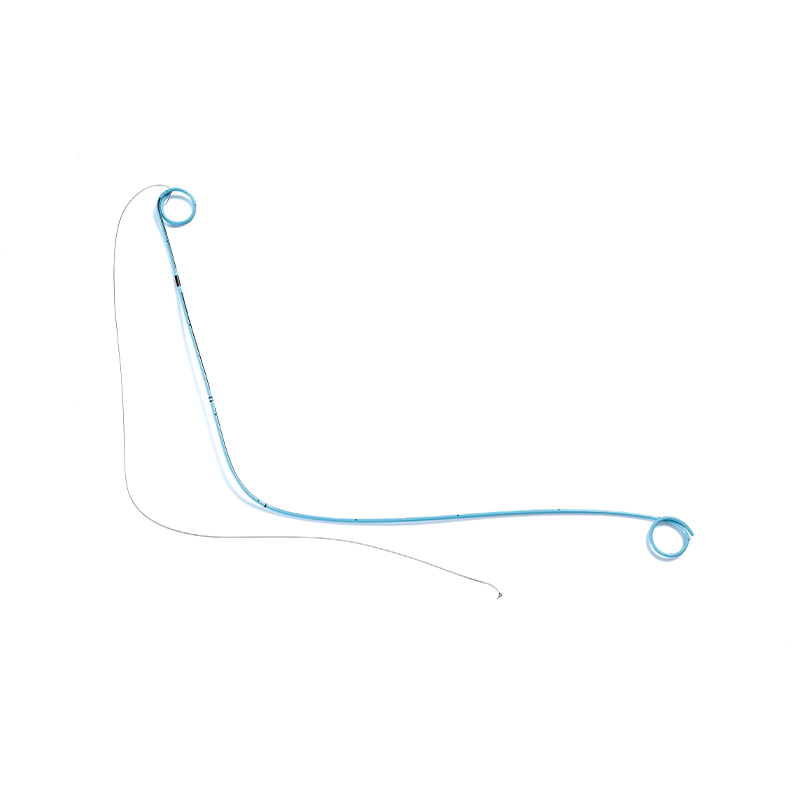
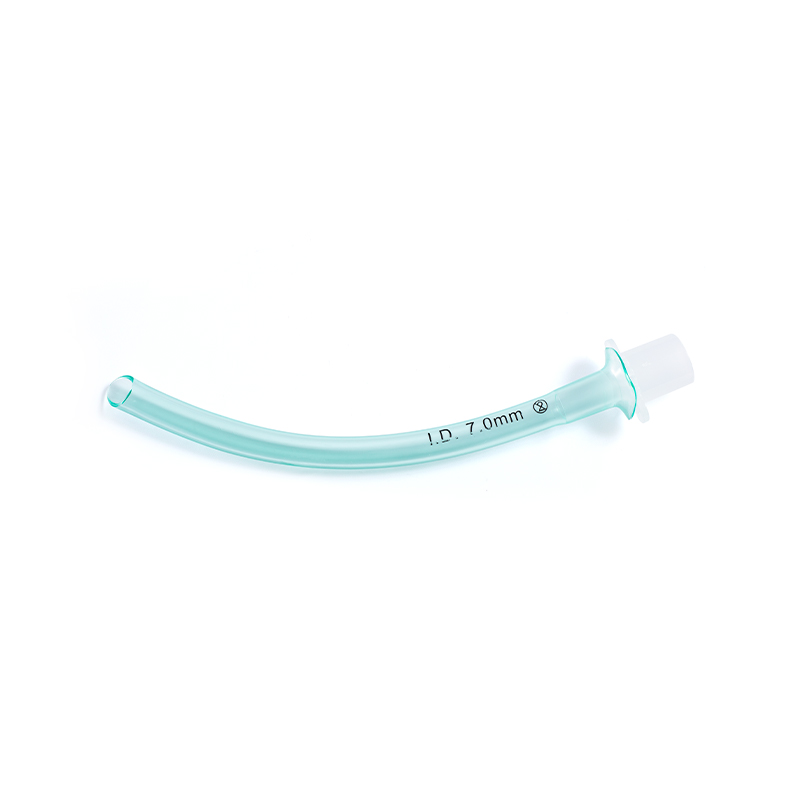
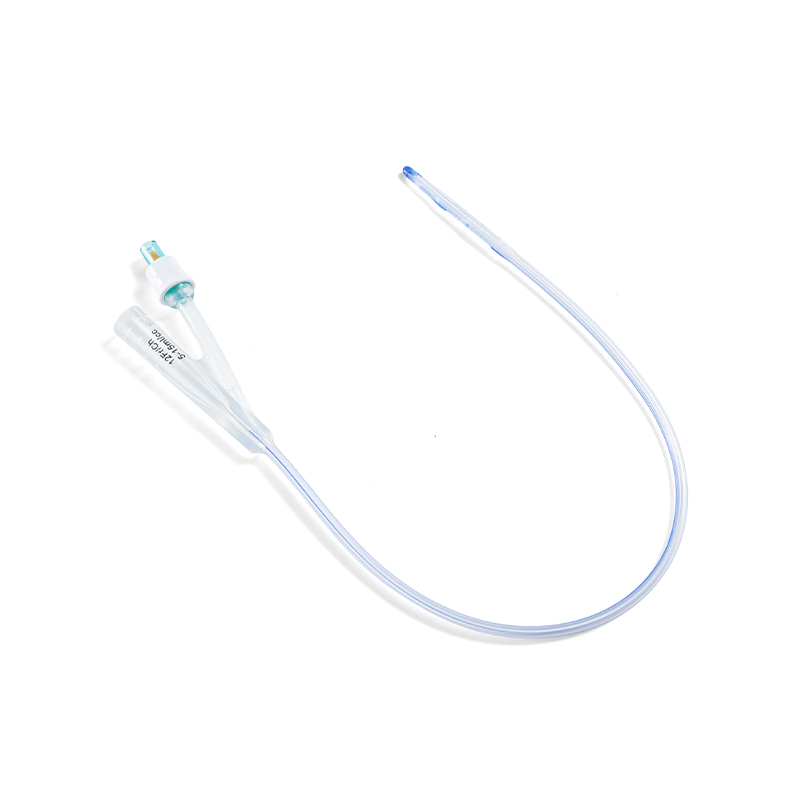
SUNGOOD, established in 2006 as a subsidiary of the Shuguang Proprietary Group, is a high-tech enterprise based in Hangzhou. Hydrophilic Coated Medical Single-Use Sterile Ureteral Stent suppliers and Hydrophilic Coated Medical Single-Use Sterile Ureteral Stent factory in China. We specialize in the development and manufacturing of high-quality disposable medical catheters, including hydrophilic coated catheters, laryngeal mask airways, and feeding tubes. custom Hydrophilic Coated Medical Single-Use Sterile Ureteral Stent. Our products are widely used in urology, gastroenterology, anesthesiology, and respiratory care.
Guided by our philosophy of “integrity, cooperation, and mutual success,” we are committed to continuous innovation and building an independent brand in the field of medical consumables in China. We strive to contribute to the medical industry and look forward to collaborating with partners to create a promising future together.
 English
English Español
Español